Why Rinse?
Airblast sprayers are not rinsed as frequently or as diligently as field sprayers. This is primarily because they are not used to spray herbicides, so residue carry-over doesn’t incur an immediately obvious penalty. The typical operator rinses prior to long-term storage or when cross contamination might cause some form of antagonism (e.g. dormant oil followed by Captan or sulfur).
Learn more about the difference between rinsing and cleaning in this article.
Aftermarket Rinsing Systems
Airblast sprayers can be outfitted with rinsing systems that permit operators to rinse quickly, easily, and dispose of dilute rinsate in rotating locations.
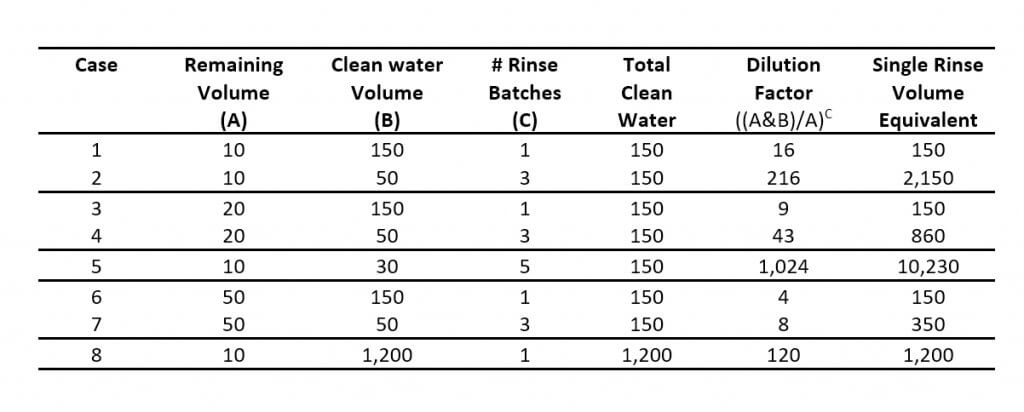
A Serial Rinse (SR) system, common on field sprayers, re-purposes the pump to transfer clean water from a saddle tank to the product tank via tank rinse nozzles. The operator introduces a volume of clean water to the remaining volume in the tank, circulates it through the system, and then sprays the rinsate in the crop. Repeating this process three times (i.e. the Triple Rinse) serially dilutes the remainder, resulting in a higher dilution factor than a single high-volume rinse.
A Continuous Rinse (CR) system requires the addition of a dedicated rinse pump. In this case the operator introduces clean water to the tank via tank rinse nozzles while simultaneously spraying. While there is circulation from the bypass (and/or agitation) circuit, the remaining volume is diluted and essentially displaced by clean water.
Objective
Using a fluorescent dye tracer as an analog for pesticide, we wanted to explore the effectiveness and efficiency of both systems. We describe the fluorimetry method in this article. We installed a CR system in a 2,000 L H.S.S. tower sprayer, which unlike most North American airblast sprayers, already features a SR system (150 L clean water tank and two tank rinse nozzles).
Installing a Continuous Rinse System
Installing a CR option required us to address the same three criteria we have already discussed in previous articles on field sprayer installs:
- Identifying a CR pump with sufficient flow to operate the tank rinse nozzles
- Satisfying the electrical or hydraulic requirements of the CR pump
- Matching the supply flow from the CR pump to the demand flow at the booms

We mounted two electric Shurflo pumps in parallel to provide flow sufficient to match the typical demand at the booms without excessive electrical load.

We found that while the CR flow spun the tank rinse nozzles weakly, the spray didn’t reach all interior surfaces. This was remedied by adding a deflector plate to the bottom of the nozzles to redirect flow.

We encountered a complication installing CR on an airblast sprayer compared to a field sprayer. Most field sprayers have rate controllers that permit the operator to adjust travel speed or ‘dial in’ a rate to match boom demand to CR pump supply. Unless the airblast sprayer already has this feature, the operator has to calculate in advance how best to match the flows.
The calculation has to be performed for each unique output (e.g. dilute or concentrate nozzle arrangements). The flow from the CR pump is a known constant. The nozzle output is variable according to operating pressure, calculated using a nozzle guide. The operator can adjust pressure (bypass or pressure regulator), PTO-speed (on positive displacement pumps), or even alternate between booms or boom-sections to match the flows.
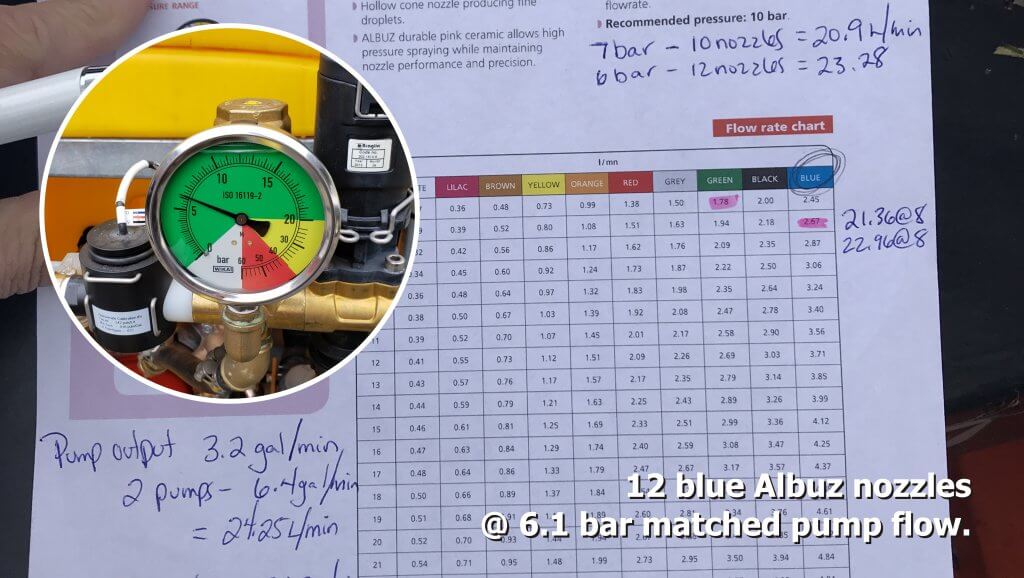
In our case, the operator was using 12 blue Albuz hollow cones in their orchard. We knew the CR pump output was 24.25 L/min. So, by setting the pressure to 6.1 bar prior to rinsing, we were spraying about 24.5 L/min. We parked the sprayer and watched to ensure the sump did not fill or drain during CR. Note in the following video how well the two flow rates were balanced (the camera was accidentally turned when we showed the vertical boom).
During trials we noticed that as the sprayer climbed uphill the water level in the tank shifted and the pump drew a little air, causing the nozzles to briefly sputter. This was a welcome sight given reports that introducing a few air bubbles during continuous rinsing can be beneficial.
Field Testing
During testing, we filled the 2,000 L Hol sprayer with 500 L of water and a final concentration of 0.25 ppm rhodamine (0.5 mL dye per 500 L water). The clean water tank was filled to 150 L. We allowed the mix to circulate for two minutes before priming the booms by spraying for a minute. A 50 mL sample was then drawn from the manifold (see below) and later used to represent the starting concentration during the analysis. The sprayer then drove through the orchard, spraying until empty.

Serial Rinse testing: When the sprayer was empty, the operator left the cab to introduce 75 L of clean water to the main tank via the tank wash nozzles. The rinsate was circulated for one minute before the operator returned to the cab and sprayed the orchard until empty. A 50 mL sample was drawn from the manifold to represent the concentration half-way through the rinse. The process was repeated for the remaining 75 L of clean water and a second 50 ml sample was drawn to represent the final concentration. We did this twice. It took about 12 minutes to rinse the sprayer and the operator had to leave the tractor cab twice.
Continuous Rinse testing: When the sprayer was empty, the operator stopped spraying and engaged the continuous rinse pump. After a few seconds, he continued driving and spraying rinsate. When 75 L had passed through the system, we paused to draw a 50 mL sample from the manifold to represent the concentration half-way through the rinse. The operator continued until the remaining 75 L was sprayed and a second 50 ml sample was drawn to represent the final concentration. We did this twice. It took about 5 minutes, 45 seconds to rinse the sprayer and the operator did not leave the tractor cab.
Sample Analysis: A Turner TD 700 fluorometer was calibrated using samples from the tank. Samples were diluted when necessary to ensure they fell in range of the calibration curve (where there is a linear relationship between the concentration of Rhodamine WT and Raw Fluorescence Units (FSU)). This range spanned a maximum of 0.1 ppm and a detection limit of 0.01 ppm active ingredient. Having previously tested recovery accuracy of 95%, data was adjusted accordingly.
Observations
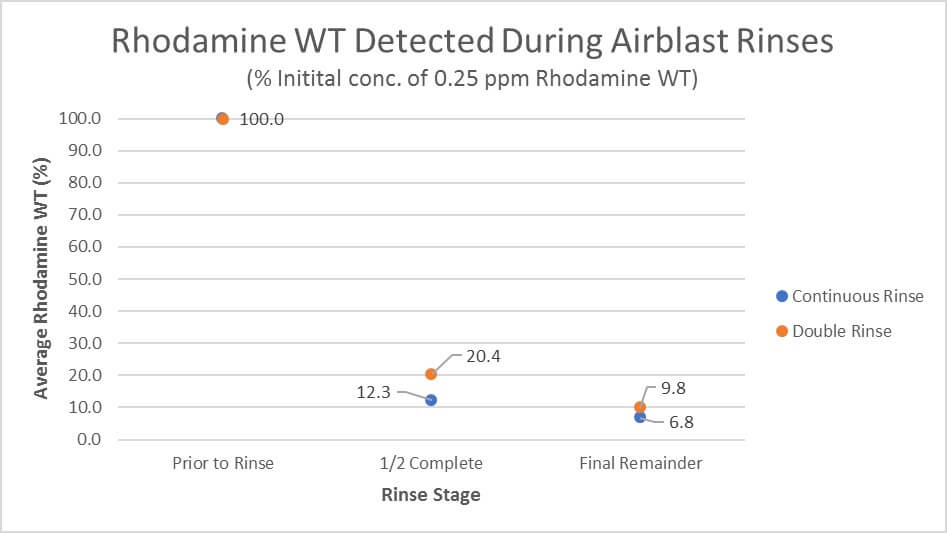
While both methods diluted the residue significantly, the remainder following both Serial and Continuous Rinse was much higher than anticipated. This may be an artifact given that both concentrations are potentially below our detection limit, per the following:
Assuming 10 L of residual spray volume left in the system once “empty”, 75 L added would give a dilution factor of 9 (according to the ). While the first 75 L of Continuous Rinse seems to remove more residue than a single addition of 75 L, both are higher than anticipated. A subsequent addition of 75 L should result in a dilution factor of 72. In this case, the remainder would be below our fluorometer’s detection limit, and could explain the results.
Nevertheless, there were positive observations:
- Continuous Rinse resulted in a more dilute rinsate with less water than Serial Rinse.
- Continuous Rinse took less time than Serial Rinse.
- The operator did not leave the tractor cab during Continuous Rinse.
- Potentially, any remaining water from the Continuous Rinse system could be used to operate a spray wand to rinse the sprayer exterior before leaving the crop.
- Both systems encourage improved airblast sprayer sanitation and reduce environmental impact from point source contamination.
Thanks to ProvideAgro for performing the installation, Wilmot Orchards in Ontario for supplying the sprayer and running the trials, and OMAFRA summer student Aidan Morgan for assistance with the data analysis.


 Figure 1: Sprayer fill station
Figure 1: Sprayer fill station Figure 2: Canada’s first commercial biobed installation at Indian Head, SK, 2009 (Source: Murray Belyk, Bayer CropScience (retired)).
Figure 2: Canada’s first commercial biobed installation at Indian Head, SK, 2009 (Source: Murray Belyk, Bayer CropScience (retired)).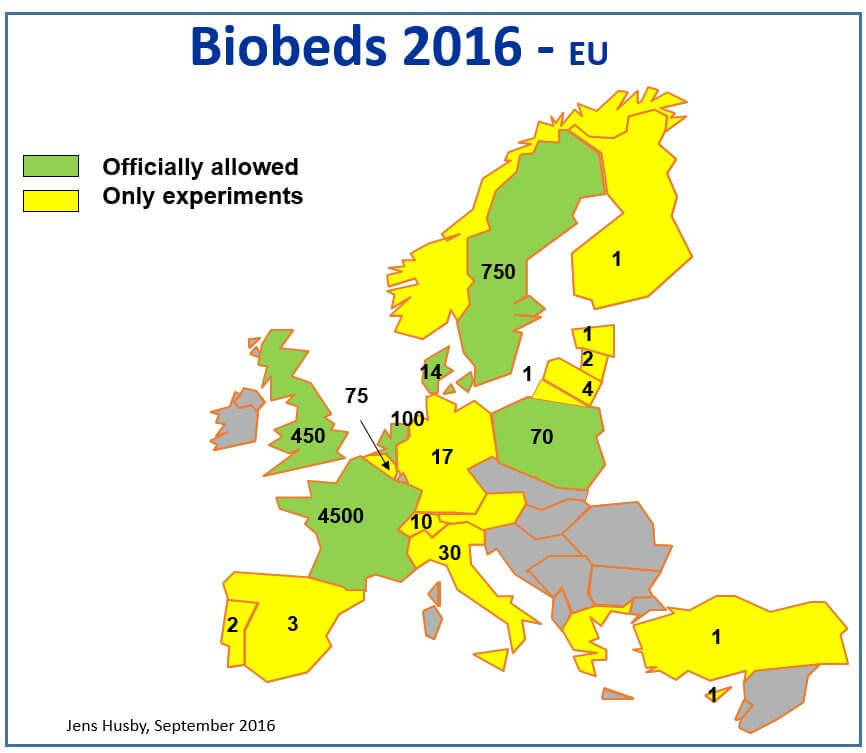 Figure 3: European biobed installations, 2016 (Source: Jens Husby, Biobeds.org).
Figure 3: European biobed installations, 2016 (Source: Jens Husby, Biobeds.org).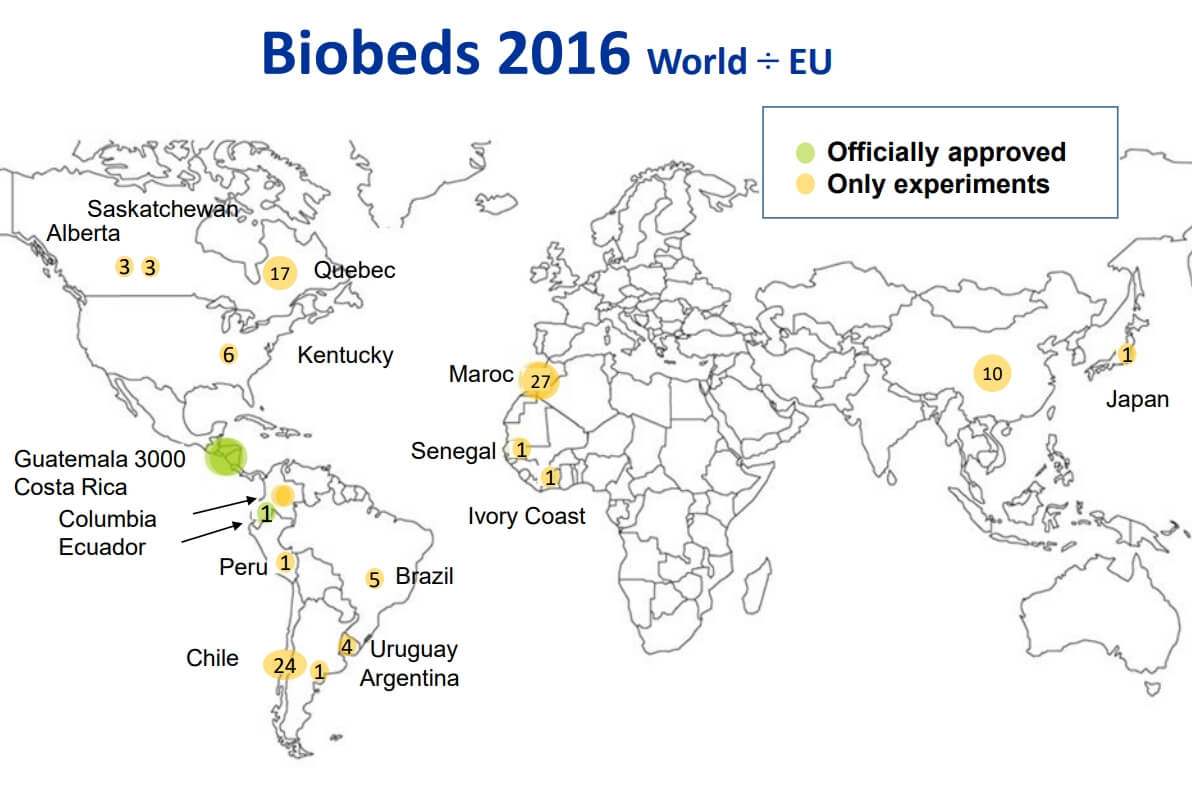 Figure 4: Global biobed installations, 2016 (Source: Jens Husby, Biobeds.org).
Figure 4: Global biobed installations, 2016 (Source: Jens Husby, Biobeds.org).
 Figure 5: Biomix preparation.
Figure 5: Biomix preparation. Figure 6: white mold (Source: AAFC).
Figure 6: white mold (Source: AAFC). Figure 7: Site selection and/or biobed covering are essential to avoid waterlogging (Source: Murray Belyk, Bayer CropScience (retired)).
Figure 7: Site selection and/or biobed covering are essential to avoid waterlogging (Source: Murray Belyk, Bayer CropScience (retired)). Figure 8: A nice looking pit.
Figure 8: A nice looking pit. Figure 9: Liner creates a closed system that will require a way to remove leached water.
Figure 9: Liner creates a closed system that will require a way to remove leached water. Figure 10: Weeping tile to collect excess water.
Figure 10: Weeping tile to collect excess water. Figure 11: Pea gravel over weeping tile.
Figure 11: Pea gravel over weeping tile. Figure 12: Filled biobed.
Figure 12: Filled biobed. Figure 13: Early sod growth on biobed at Indian Head, SK.
Figure 13: Early sod growth on biobed at Indian Head, SK. Figure 14: Pesticide waste entering biobed via drip irrigation.
Figure 14: Pesticide waste entering biobed via drip irrigation. Figure 15: Biobed system in Simpson, SK. Rinsate from sprayer is collected in a sump, which is pumped to the black storage tank in background. Rinsate is introduced into biobed (blue tub) as needed (Brian Caldwell in foreground, left, Larry Braul, right).
Figure 15: Biobed system in Simpson, SK. Rinsate from sprayer is collected in a sump, which is pumped to the black storage tank in background. Rinsate is introduced into biobed (blue tub) as needed (Brian Caldwell in foreground, left, Larry Braul, right). Figure 16: Holding tank at biobed in Outlook, SK.
Figure 16: Holding tank at biobed in Outlook, SK. Figure 17: Steel beams can allow (light) sprayer access (Source: Eskil Nilsson via Biobeds.org).
Figure 17: Steel beams can allow (light) sprayer access (Source: Eskil Nilsson via Biobeds.org). Figure 18: Two-stage biobed system at Outlook, SK.
Figure 18: Two-stage biobed system at Outlook, SK. Figure 19: Above ground biobed installation with plastic tub.
Figure 19: Above ground biobed installation with plastic tub. Figure 20: Heat tape (Source: AAFC).
Figure 20: Heat tape (Source: AAFC). Figure 21: Passive solar biomix heating system.
Figure 21: Passive solar biomix heating system.
 Figure 22: Phytobac installation, cross-section.
Figure 22: Phytobac installation, cross-section. Figure 23: Biofilter installation in Belgium (Source: Inge Mestdagh via Biobeds.org).
Figure 23: Biofilter installation in Belgium (Source: Inge Mestdagh via Biobeds.org).