In June 2013 we ran a ginseng spraying workshop and we learned as much as the growers did. Ginseng is notoriously difficult to spray:
- It is highly susceptible to pathogens given the high humidity and still conditions generally found under the shade structure.
- It forms a solid ceiling of leaves that resist spray penetrating to the stem and crown below and makes under-leaf coverage very difficult to achieve.
Many growers have (wisely) walked away from the old Casotti sprayers, which have been shown to give erratic coverage at best. They have adopted the Arag Microjet system with it’s characteristic orange shields. The >$80.00 CAD price tag for each nozzle is due to the brass mixing valve and swivel joint, as well as import costs from Italy. Contrary to popular belief, it does not use air-assist, or air-induction – it is strictly hydraulic. It does tend to create a ‘wake’ of air movement at high pressure. This phenomenon is called air entrainment and it is caused by large droplets travelling at high speed.

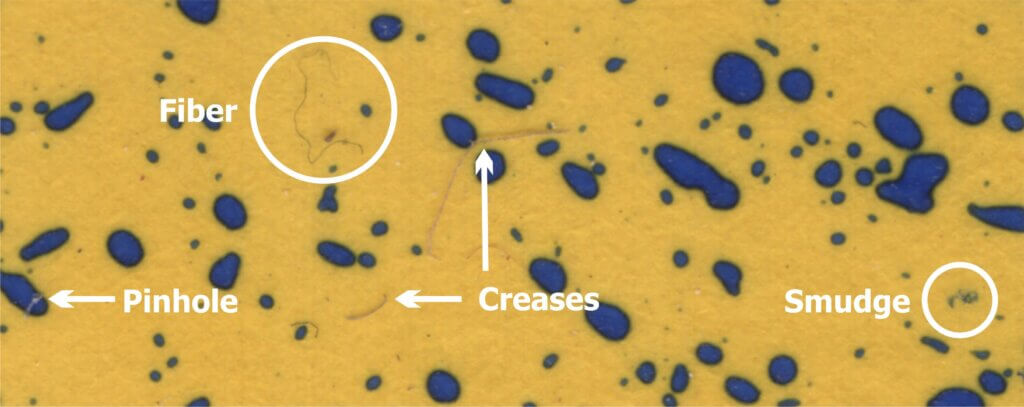
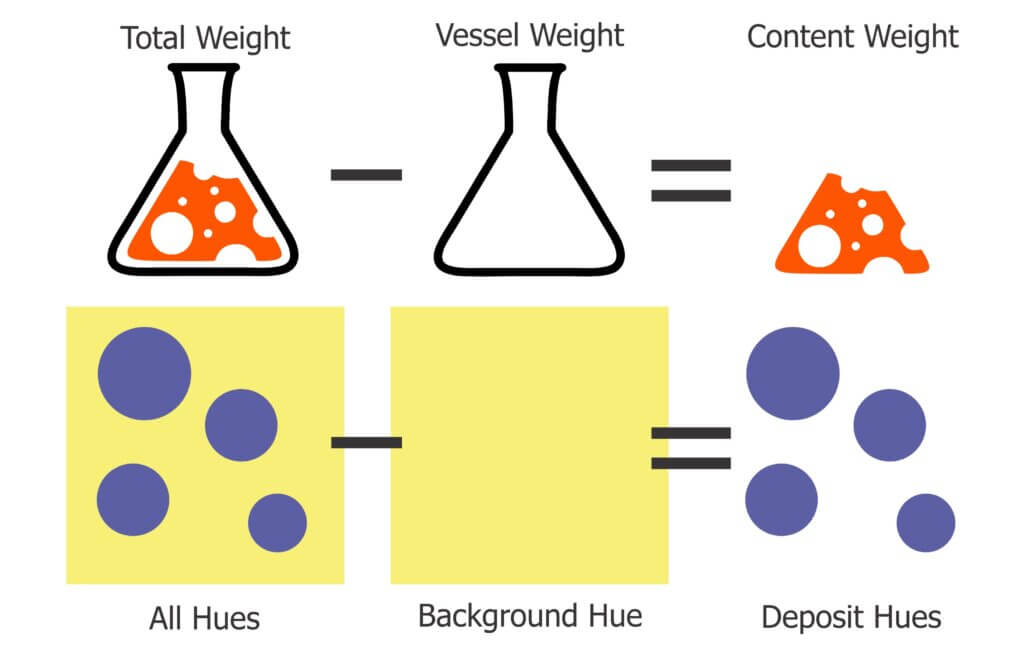
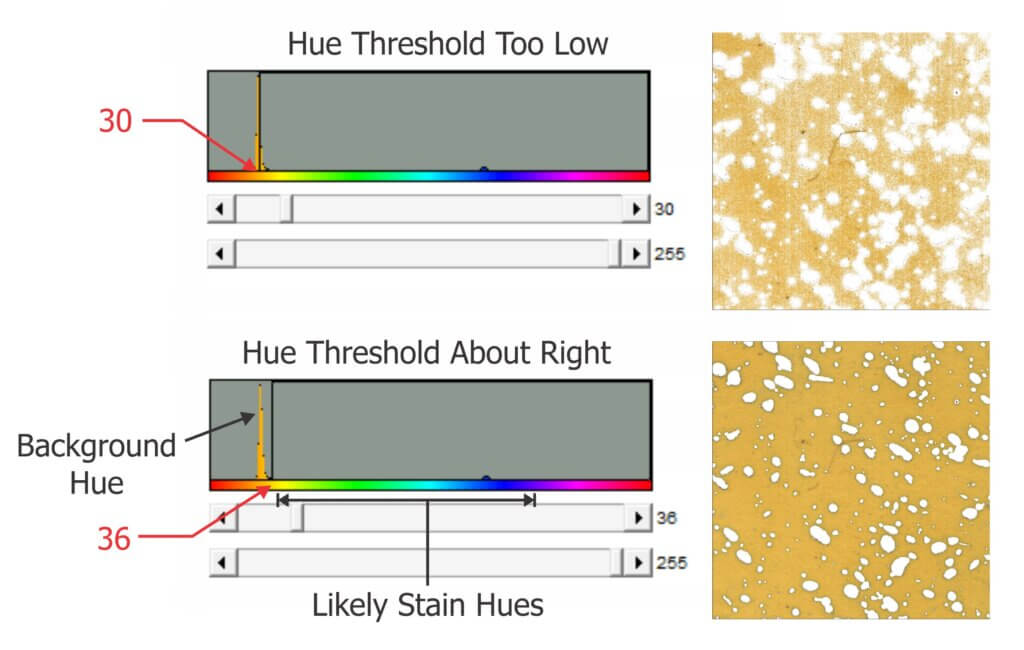
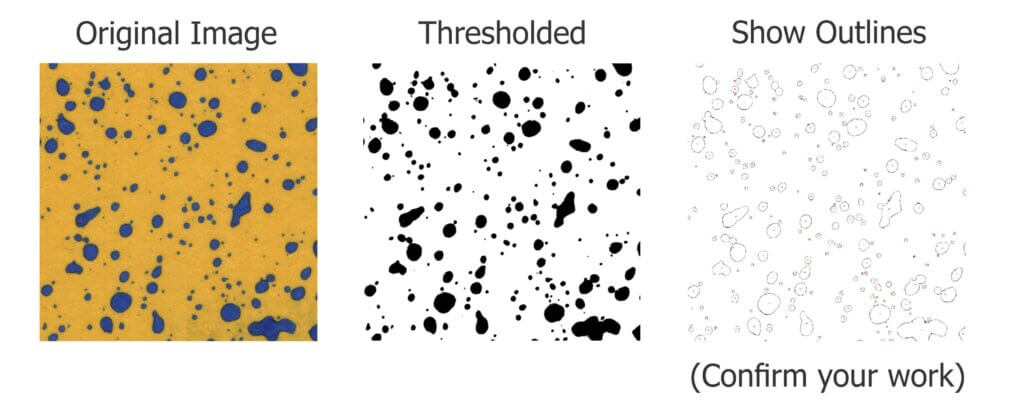
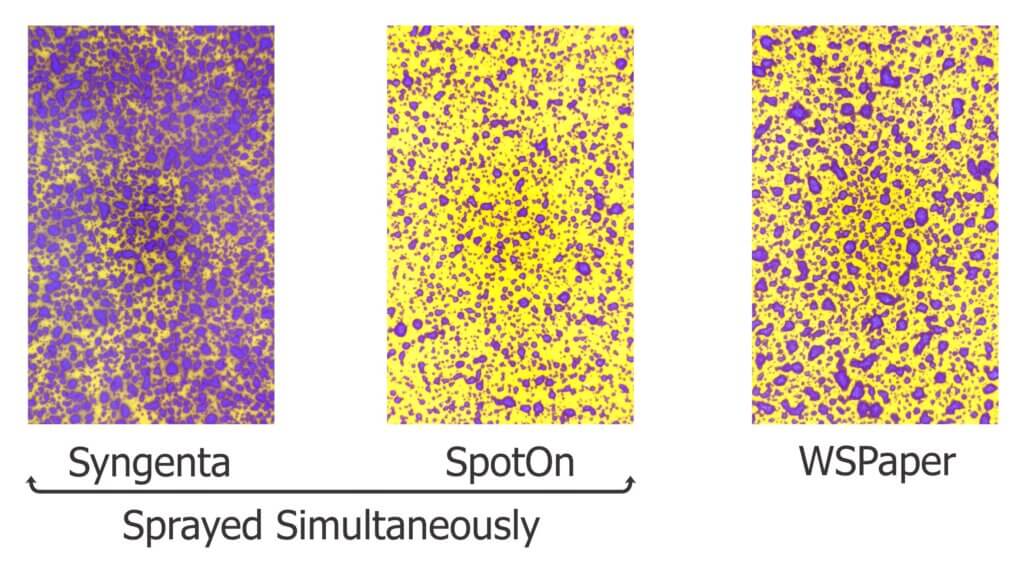
This nozzle is essentially the business-end of a spray gun. The way it is used in ginseng it works more-or-less like a hollow cone disc-core assembly. This begs the question “Why not use the cheaper and more readily available ceramic disc-core?” We set out to compare the two options using water sensitive paper set within the canopy. These yellow, paper targets turn blue when sprayed, clearly showing spray coverage.
Determining rates
The first step was to determine the output rate for each nozzle. Generally, nozzle manufacturers provide rate tables showing how much volume a nozzle emits by time (e.g. US gallons per minute) at a given pressure. Finding these tables for the 1.5 millimetre Arag Microjet proved difficult. When we finally found one, it was discovered the rates were established for 200 to 850 pounds per square inch. This is excessively high pressure for a typical boom sprayer, so tables had to be developed for lower pressures.


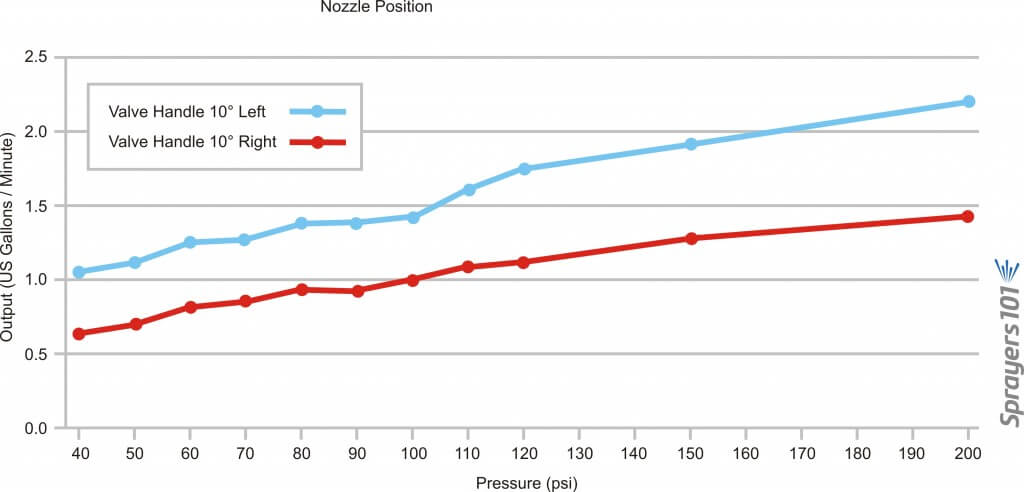
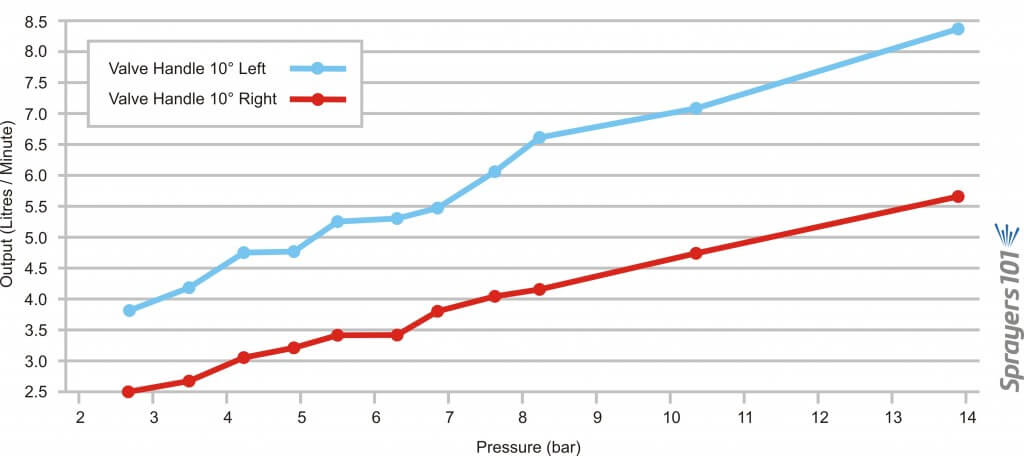
Further, given the odd design of the mixing valve, it was determined that moving the handle ~10 degrees left of centre, or ~10 degrees right of centre, gave a difference of as much as 60%. The table below shows the outputs for a 1.5 millimetre nozzle with the handle in both positions and the two graphs show the results… well… graphically. Outputs were determined using the Innoquest Spot-On SC-4, but the frothing effect created by the nozzles may have created minor errors. Each rate is the average of a minimum of three samples.
| Valve Setting | Pressure (psi) | Avg Output (gpm) | Pressure (bar) | Avg Output (L/min) |
| 10 degrees left | 40 | 1.02 | 2.76 | 3.86 |
| 10 degrees left | 50 | 1.1 | 3.45 | 4.16 |
| 10 degrees left | 60 | 1.25 | 4.14 | 4.73 |
| 10 degrees left | 70 | 1.25 | 4.83 | 4.73 |
| 10 degrees left | 80 | 1.38 | 5.52 | 5.22 |
| 10 degrees left | 90 | 1.4 | 6.21 | 5.3 |
| 10 degrees left | 100 | 1.45 | 6.89 | 5.49 |
| 10 degrees left | 110 | 1.6 | 7.58 | 6.06 |
| 10 degrees left | 120 | 1.75 | 8.27 | 6.62 |
| 10 degrees left | 150 | 1.87 | 10.34 | 7.08 |
| 10 degrees left | 200 | 2.2 | 13.79 | 8.33 |
| 10 degrees right | 40 | 0.65 | 2.76 | 2.46 |
| 10 degrees right | 50 | 0.7 | 3.45 | 2.65 |
| 10 degrees right | 60 | 0.8 | 4.14 | 3.03 |
| 10 degrees right | 70 | 0.85 | 4.83 | 3.22 |
| 10 degrees right | 80 | 0.9 | 5.52 | 3.41 |
| 10 degrees right | 90 | 0.9 | 6.21 | 3.41 |
| 10 degrees right | 100 | 1 | 6.89 | 3.79 |
| 10 degrees right | 110 | 1.07 | 7.58 | 4.05 |
| 10 degrees right | 120 | 1.1 | 8.27 | 4.16 |
| 10 degrees right | 150 | 1.25 | 10.34 | 4.73 |
| 10 degrees right | 200 | 1.37 | 13.79 | 5.19 |
Comparing nozzles
Using the grower’s typical ground speed of 5 km/h (~3 mph) and operating pressure of 6.9 bar (100 psi), we found four TeeJet disc-core combinations that emitted a hollow cone pattern and approximately the same output as the Arag Microjets. The five nozzles sets tested were:
- ARAG Microjet® 1.5 mm = ~0.95 US g/min avg at 100 psi
- TeeJet® D8-DC25= 0.97 US g/min at 100 psi= ~97° cone
- TeeJet®D7-DC45= 0.97 US g/min at 100 psi= ~81° cone
- TeeJet®D4-DC46= 0.88 US g/min at 100 psi= ~33° cone
- TeeJet®D6-DC45= 0.93 US g/min at 100 psi= ~81° cone
We did not use nozzle drop hoses (aka drop arms or hose drops) because it has already been firmly established that they are absolutely required to achieve under leaf coverage See OMAFRA factsheet 10-079 and this article.
Observations
While there were some complications with setting up the papers for the demo, we observed the following:
- The output of each Microjet nozzle can be as much as 50% more or less than expected without being visually detectable and output for each nozzle must be confirmed before spraying. Therefore, outputs should be confirmed before every application.
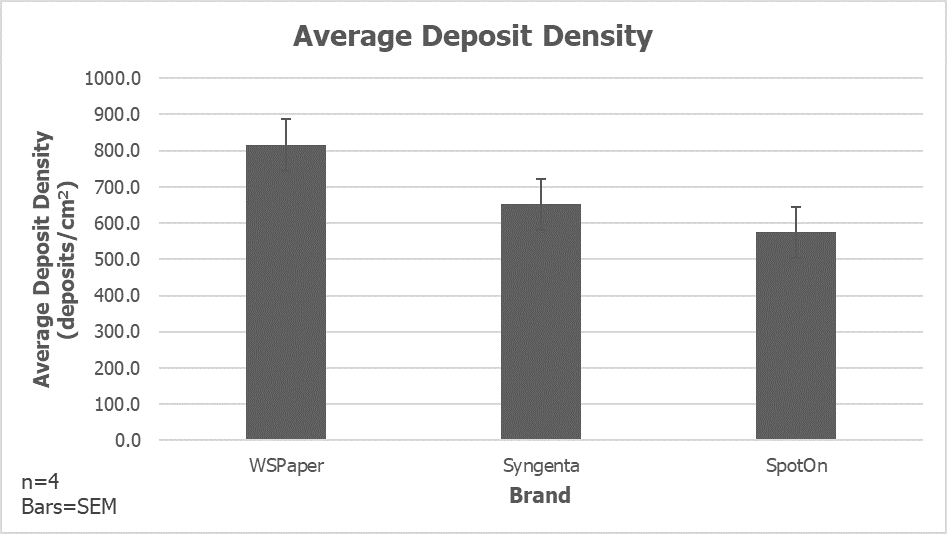
- Microjets at 100 psi emitting ~890 L/ha (~95 US gallons per acre) gave satisfactory coverage on all upward facing targets, but unsatisfactory under-leaf coverage. This has been demonstrated many times before.
- The TeeJet D7-DC45 combination emitting a similar rate gave satisfactory coverage on all upward facing targets, but unsatisfactory under-leaf coverage. They may be a viable alternative to the Microjets.
- Nozzle drops are advised to achieve under-leaf coverage.
The demo also raised some questions:
- Did the TeeJet disc-core push the canopy apart as much as the Microjet? The audience noticed there was some leaf-shadowing where the cards did not get complete coverage using disc-core. This might have been coincidence, or it may not have. This question will be addressed in a research trial next season, but for now, the D7-DC45 appeared to give similar coverage to the Microjet.
- Can nozzle drops be avoided if pressure is raised to 27.5 bar (400 psi)? Thanks to one grower trying this experiment in his garden after the demo, we saw some under-leaf coverage is possible at such high pressures, but this occurred at the cost of a lot of noise, diesel fuel and considerable wear on the ceramic Microjet discs. The grower tested these tips and discovered they needed replacement after only two years of use. Nozzle drops are cheaper, easier and result in considerably more spray in the under leaf positions.
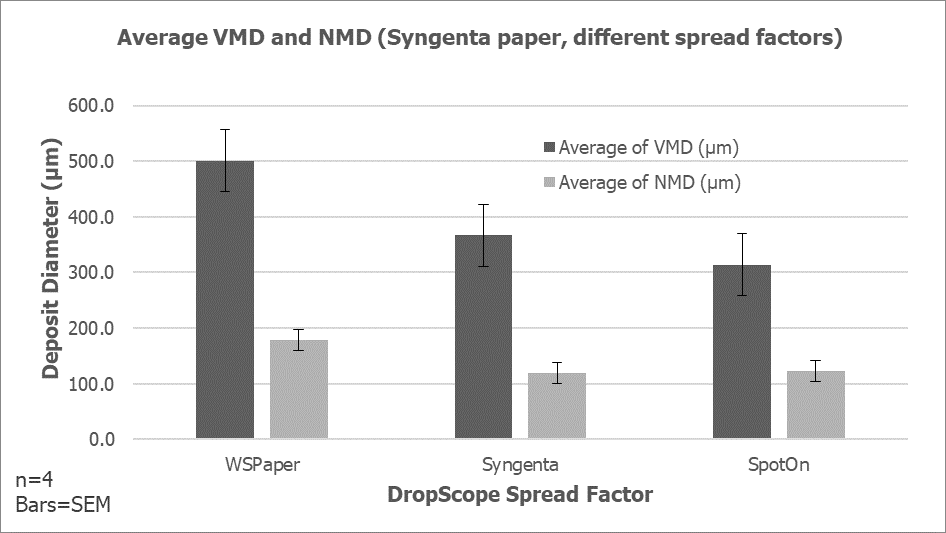
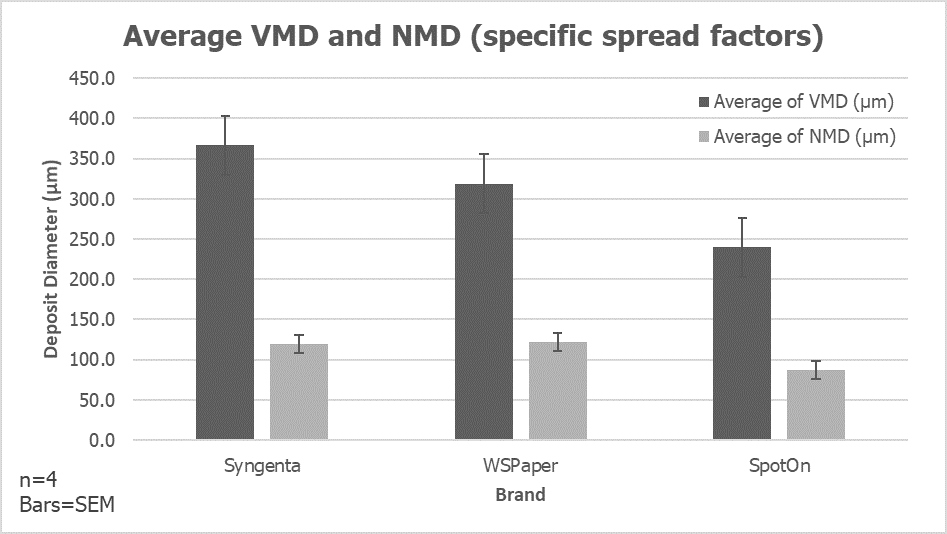
- We saw what minimal and excessive foliar coverage looked like, and determined how much variability there was from one nozzle to another. A significant question was “How much spray can be saved when using a more accurate application?” and the answer is yet to be determined, but could be well in excess of 10% of the typical spray volume. Given that this crop can be sprayed more than 100 times over it’s 3 or four years before harvest, this represents significant savings in pesticides and refill time.
Additional – Newer ARAG Microjet Design
Since this work was performed, growers have been exploring a newer option from ARAG.
They are an improvement over the older version insofar as they are more easily calibrated and held at a given rate thanks to a lock nut. They still employ a 1.5 mm diameter ceramic disc, but this can be changed for a 1.0 or 1.2 quite easily. They are still somewhat finicky when trying to set a consistent spray quality and rate from nozzle to nozzle, but are better than the mixing-valve option.
Learn more in this article.

Special thanks to Syngenta Canada for providing lunch, to C&R Atkinson Farms Ltd. for hosting, to TeeJet for supplying the disc-cores and water-sensitive papers, and to Dr. Sean Westerveld, Dr. Melanie Filotas and OMAFRA summer student Megan Leedham for contributing to the workshop.