If you’re a sprayer operator with some experience behind you, you may have applied mercury arsenate, nicotine, Paris green, or perhaps even DDT. All of these historical pesticides were effective, but they were also toxic to both the applicators and the environment. Fortunately, today’s agrochemical manufacturers produce pesticides that are effective while being far less hazardous.
One important aspect of modern synthetic pesticides that enhances their efficacy is their ability to redistribute. Pesticide redistribution is the movement of a pesticide from its initial point of deposition to a different spot on or in the plant. Pesticides that can redistribute can improve pest control compared to those that must contact the target pest but cannot innately redistribute. This is especially true when spraying hard-to-wet plant tissues, such as flower clusters or fruit. Even when the immediate coverage of these tissues is insufficient, the subsequent relocation beyond the initial spray deposit can result in a more effective protective barrier. When plants are rapidly growing, many of these products can translocate through the plant tissues to protect newly emerged tissue that did not receive a direct deposit.
Some of the most difficult and persistent pests are more effectively controlled by redistributing pesticides. Materials that move within the plant after application provide improved control of piercing-sucking insects such as aphids and psyllids, as well as pests that feed in difficult-to-spray areas such as under leaves. These products can absorb into plant tissue, increasing their resistance to wash-off by rain or irrigation.
Five Types of Pesticide Redistribution
There are five significant types of pesticide redistribution: translaminar, vapor, xylem, phloem and redistribution via precipitation
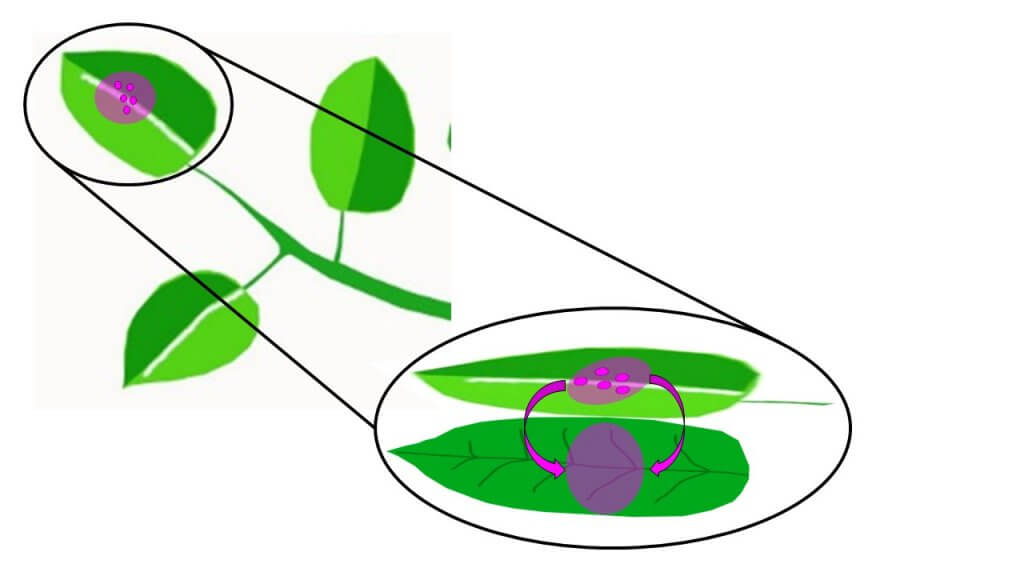
Translaminar Redistribution
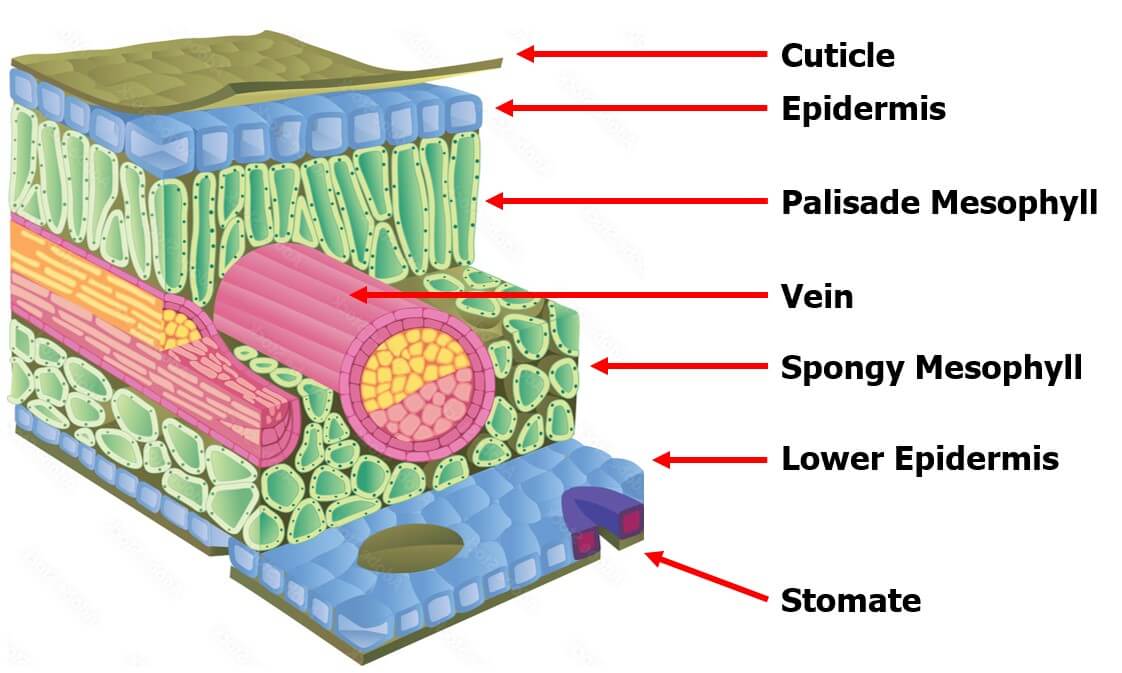
Translaminar redistribution (Figure 1) in its most literal sense is a compound moving from the side of the leaf that received spray, to the unsprayed opposite side. This results in protection on both sides. However, translaminar redistribution also involves limited radial movement providing a “halo” of protection around the initial deposition. The extent of this area of influence is product-dependent.
Vapor Redistribution
Vapor redistribution (Figure 2A) occurs when surface depositions volatilize and move laterally along a plant surface, re-adsorbing to the plant surface in new locations as they move. Again, the extent of vapor activity is product specific, but also condition specific requiring an optimal combination of temperature, relative humidity, wind, and solar radiation to facilitate volatilization. When pesticides are referred to as “locally systemic,” it often implies that they exhibit translaminar and/or vapor redistribution properties.
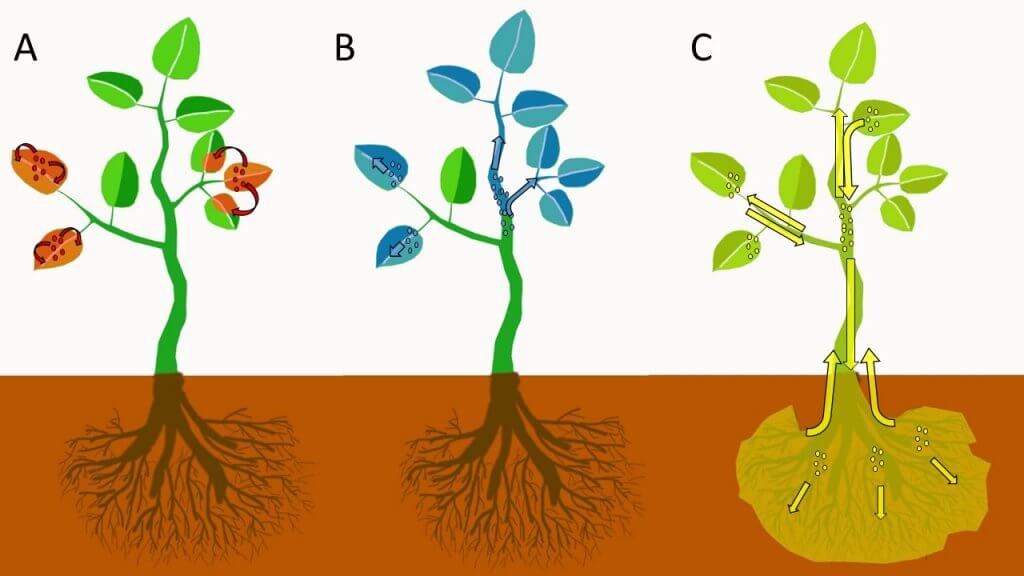
Xylem and Phloem Redistribution
Xylem redistribution (Figure 2B), also called xylem systemic, refers to the absorption of a pesticide and subsequent systemic movement of the pesticide through the xylem vessels of a plant. Xylem vessels move water and minerals in an upward and outward direction in plants. There is very little movement of water and nutrients downwards or backwards along branches or leaves in xylem vessels. Xylem redistribution can help protect growing tissues from damage by pests or diseases when the pesticide redistributes from the point of application to the newly developing tissues. Most systemic fungicides and insecticides redistribute via the xylem.
Phloem redistribution (Figure 2C), also called phloem systemic, is the bi-directional movement of pesticides in the phloem vessels of a plant. Phloem vessels transport sugars and other nutrients both to the roots of plants and upwards and outwards to shoots and fruits/seeds. Phloem systemic pesticides are sometimes called “true systemic,” because they can translocate throughout the entire plant.
Some pesticides that redistribute via the xylem or phloem can be applied to the soil substrate to be absorbed by the roots and redistributed throughout the plant. The process of plant nutrients or pesticides being transported from one place to another within the plant is called translocation.
Soil-Applied Systemics
Several factors affect a pesticide’s ability to redistribute. These factors affect the speed of uptake, the duration and extent of translocation, and the amount of accumulation in plant tissue relative to the initial dose. For pesticides labeled for soil application, their uptake by plant roots and redistribution via xylem or phloem can lead to long residual efficacy of the product; Up to eight weeks or more depending on the product, plant, and soil. This is in contrast to foliar-applied products, where good residual efficacy could be expected to last two to three weeks depending on the product. However, foliar-applied products tend to provide a more rapid kill of target pests and a more rapid absorption and translocation of active ingredients.
For soil-applied systemic pesticides, the composition of the soil substrate can affect the uptake of the pesticide by the plant. Growing media high in organic matter (>30% bark or peat moss) can bind pesticides, making it difficult for plants to absorb them through roots and subsequently translocate via the plants vascular system. Soil applications of systemic materials should take place one to six weeks prior to the onset of the insect pest or pathogen. This allows sufficient time for the pesticide to translocate to, and accumulate in, target tissues. The more water-soluble pesticides (e.g. Thiamethoxam) are taken up more rapidly than the less water-soluble pesticides (e.g. Imidacloprid).
Redistribution via Precipitation
In contrast to systemic pesticides, contact pesticides cannot redistribute on their own. However, rain or irrigation can spread the deposit to some degree, increasing coverage area. This effect should not be relied upon, as it depends on the product formulation, the intensity of the precipitation, and the interval following application. In the case of prolonged precipitation, the residual activity of contact products can be greatly reduced as they are diluted and washed off plant tissues.
Plant Morphology
The status of the plant to which they are being applied is a significant consideration when applying redistributing pesticides. Both soil-applied and foliar-applied pesticides are more rapidly absorbed and redistributed when applied to young plants or juvenile plant tissue. In general, when plants are actively growing, have a strong root system, or are actively transpiring, they tend to absorb and translocate pesticides more rapidly than when plants are growing slowly. In addition, plants with difficult to wet leaves or surfaces due to thick cuticles or waxy layers tend to not absorb pesticides as readily. Penetration into plants with difficult to wet surfaces can be improved by adding adjuvants such as surfactants to tank mixes.
Multiple Modes of Redistribution
The extent to which each product can redistribute can be thought of as a continuum. Generally, when a product exhibits some form of redistribution, it can also redistribute via a different method. A good example of this is xylem and translaminar redistribution. When a product can redistribute via the xylem it generally can move through the leaf via the translaminar pathway as well. Some products can redistribute via the xylem, translaminar, and vapor pathways all at the same time. Others, while technically able to redistribute via more than one mechanism, are only biologically effective via one mechanism.
Consult the Pesticide Label and Other Reputable Sources
The best way to determine how a pesticide product redistributes is to consult the manufacturer’s label, as well as technical information from reputable sources such as government or academia. If a manufacturer provides a technical information bulletin it is generally available on their website on the pesticide product page along with the label. However, because there are no standardized metrics to rate pesticide redistribution, there can be significant disparity between products. Some products that are advertised as being xylem systemic for example, are actually less systemic than products that are not even advertised as being systemic. Additional information on the efficacy and redistributing characteristics of specific products can be obtained from extension agents or crop consultants.
Conclusion
In summary, when selecting a pesticide remember to consider the four different pathways of redistribution (xylem, phloem, translaminar, and vapor) and how these methods may improve the efficacy of your application, allowing you to get more out of every drop.