In 2016, an asparagus grower in southern Ontario picked up a used De Cloet Hi-Boy originally used to spray tobacco. His vision was to create a three-row herbicide sprayer for asparagus and we were invited to participate. His concept was to design shrouds that would contain the herbicide, but not snag the asparagus or drag heavily on the ground. This article follows the development of the sprayer from concept to testing to final product.

The sprayer itself was a classic three-wheel, self-propelled affair. The asparagus was planted on four foot centres, leaving a three foot alley. While the goal was to hang three shrouds off the boom, we started with one to work out the bugs.

This operation uses 2,4-D to control weeds in the alleys and while a little can hit the asparagus stem up to 12 inches (where the branching starts), we wanted to avoid contact at all costs. That led us to the TeeJet AI 95° flat fan nozzle, which produces a Very Coarse to Extremely Coarse spray quality. A single nozzle could be suspended to span the 3 foot width of the alley.


The first version of the shroud was suspended off the boom from four anchorage points. A certain amount of of play was allowed so the shroud would find plumb (i.e. hang vertically), even when the sprayer boom yawed or pitched over uneven ground.
The shroud was constructed of sheet metal, angled to reduce the potential for contact with the asparagus branches, and terminated in stiff, nylon brush-style mud flaps commonly seen on trucks. These brushes were cut to a few inches in length to span the distance between the side of the shroud and the ground. This would create a “seal” to prevent spray from escaping, maintaining some degree of contact with uneven ground.

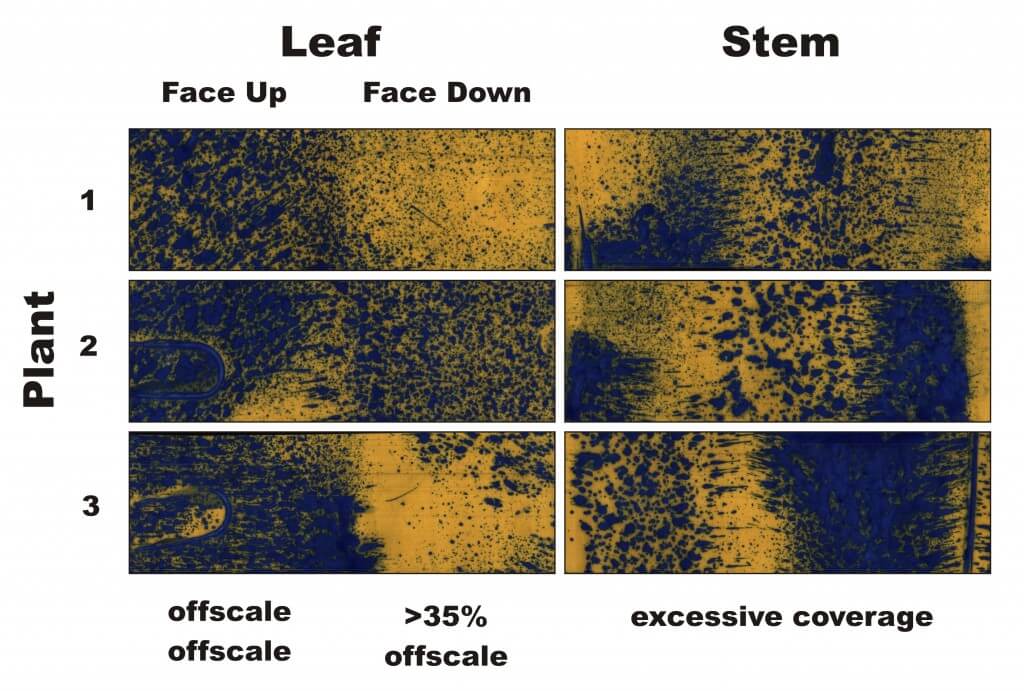
We tested the first version by placing water sensitive paper in two positions on the ground, just inside the reach of the brushes. We had to be careful not to run them over with the centre wheel of the sprayer. We also adhered two papers to the angled inner walls to see how much, if any, spray was hitting the inside of the shroud.
Our first pass on June 16th was at 9:00 am, 19.1 ºC (66.4 ºF) with a cross wind of 5 to 7 km/h (3.1 – 4.3 mph). relative humidity was high at 85% and travel speed was slow at 3.2 km/h (2 mph). We started with the .06 AI tip at 50 psi, but we drenched all the targets with excessive coverage because we were travelling so slow. We also found the stiff brushes were creating furrows in the soil, as shown below.

For our second pass, we tried the .04 tip and raised the shroud while dropping the tip to keep it suspended 15 inches over the ground. We were still drenching the targets and noticed the shroud was hitting the asparagus spears, causing physical damage. The damage is shown below – note the dark green on the bent spear.

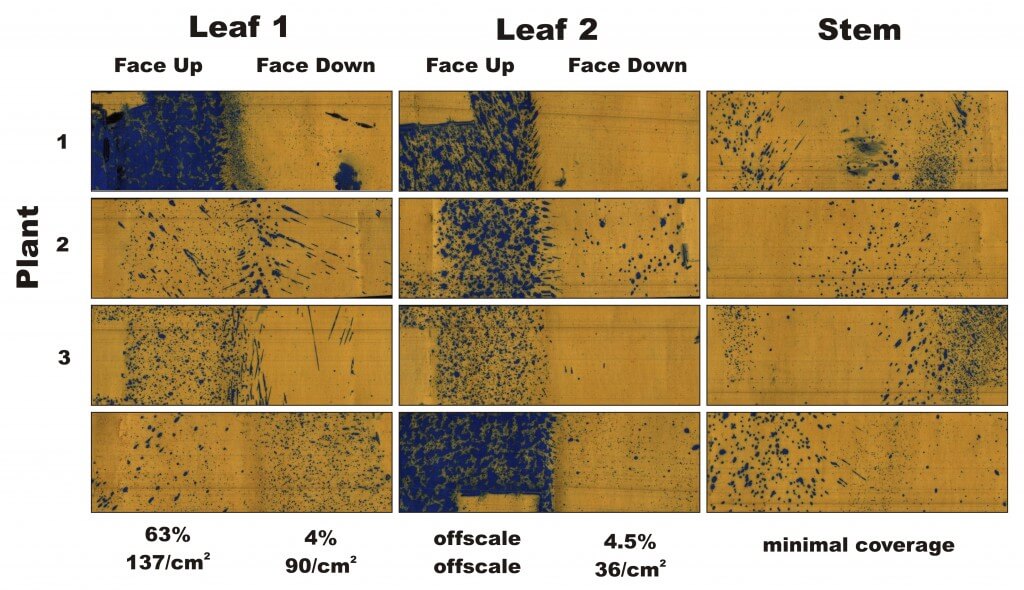
This led to a decision to flare the side walls more aggressively, bringing them further into the centre of the alley and away from the spears (shown later in the article). This had the added benefit of angling the brushes as well to get a maximum span for weed control in the alley. For the final coverage pass we used the AI .03 tip, which gave more than 45% coverage on the ground, with even distribution, and there was no indication of spray on the papers adhered to the inside of the shroud. This coverage is more than is likely required, and the operator should be able to spray up to 6.5 km/h (4 mph) without compromising coverage.


Since the coverage tests, the grower added additional sheet metal fenders to the the existing fenders, encasing the wheels and creating a smooth transition for the shroud to gently deflect the asparagus. The fenders were needed because the grower found the asparagus was being pushed out by the wheel fender only to bounce back in front of the shroud, which snagged the fern and damaged it. The additional fenders keep the fern spread and prevent it getting caught in front of the shrouds.



The grower was very happy with the sprayer’s performance and planed to build another. Why be satisfied with the status quo when you can tap into your creative side and be innovative? If you don’t think you’re imaginative enough to try upgrading equipment on your farm, here’s a simple test to prove that it’s in you. It’s easy to see the bird in the image below, but with a little concentration you’ll be rewarded with a ski-jumping rabbit.

Thanks to TeeJet for donating the nozzles and water-sensitive paper and to Ray and Brad Vogel of Lingwood Farms for inviting me to participate.
Learn more about spraying asparagus here.